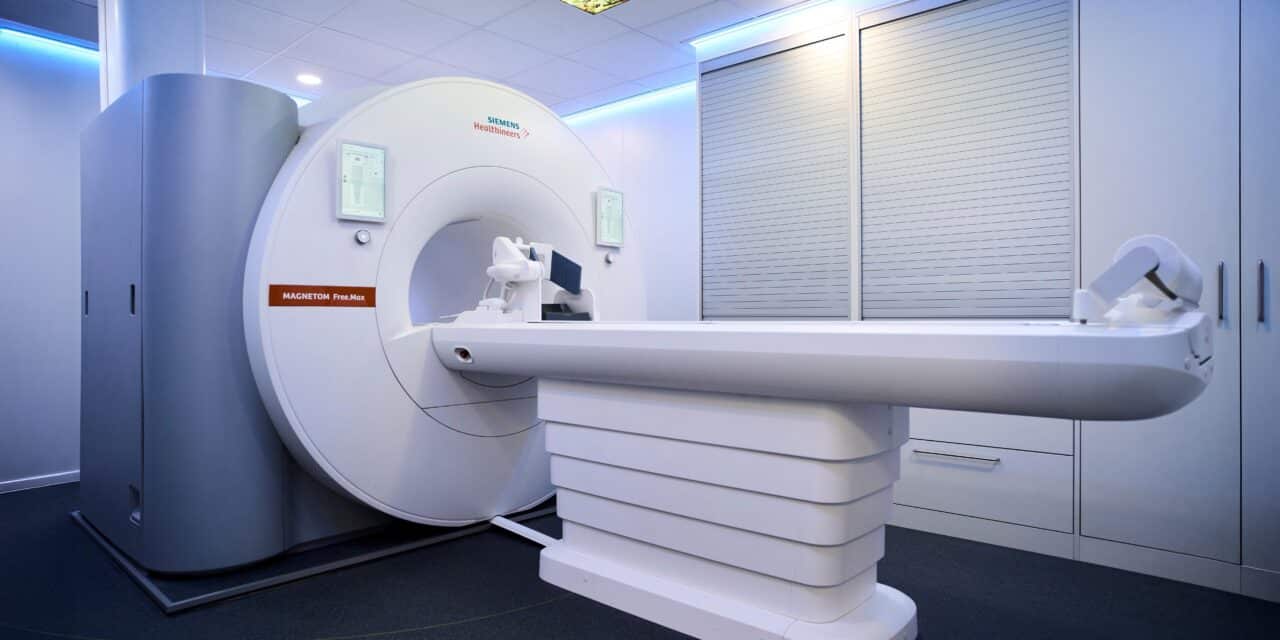
The MAGNETOM Free.Max Dental Edition is designed to provide non-ionizing soft-tissue imaging to support diagnosis and treatment planning.
Dentsply Sirona and Siemens Healthineers have received FDA clearance for their dental-dedicated MRI (ddMRI) system, the MAGNETOM Free.Max Dental Edition. The clearance follows a clinical trial that validated the system’s application in orthodontics, endodontics, periodontics, TMJ, and tooth extraction by providing non-ionizing imaging for soft-tissue diagnostics.
System Features and Clinical Use
The MAGNETOM Free.Max Dental Edition uses a dedicated dental coil and specialized software to create a dental-specific field of view that focuses on dental structures without imaging brain tissue. According to the companies, the system has an average workflow time of under 20 minutes.
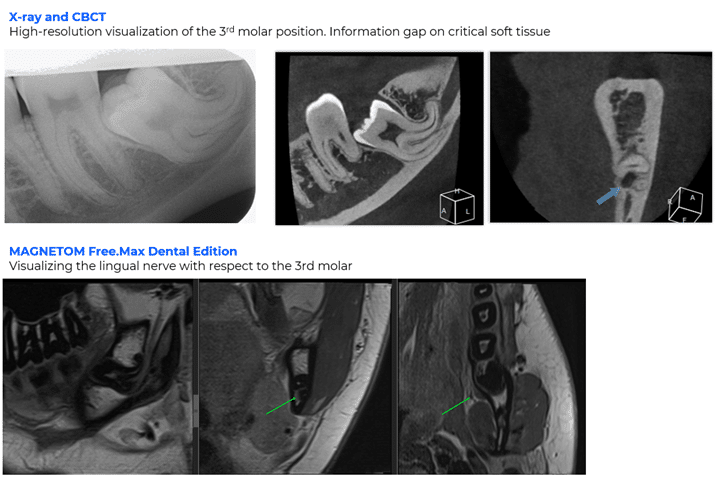
Findings from the clinical trial showed the system’s ability to differentiate between active inflammation and healthy or scar tissue, perform non-invasive assessments of tooth pulp vitality, and visualize the position of teeth in relation to neighboring teeth and nerves.
“This capacity to see the clarity of the soft tissue detail produced by ddMRI has clear potential to enhance diagnostic confidence in areas where current modalities have limitations,” says Don Tyndall, DDS, MSPH, PhD, professor of diagnostic sciences at the University of North Carolina, who reviewed the clinical trial findings.
A Complement to Existing Imaging
As an imaging modality free of ionizing radiation, ddMRI is designed to complement intraoral, panoramic, and CBCT imaging, particularly for diagnostic questions where soft-tissue characterization is needed. The system was developed for use in hospitals, large clinics, and universities.
“This innovation is a powerful addition to our imaging portfolio,” says Dan Scavilla, president and CEO of Dentsply Sirona, in a release. “We’re proud to have contributed to developing the industry’s dental dedicated MRI and the advancement of diagnostic capabilities in the field.”
Andreas Schneck, head of magnetic resonance at Siemens Healthineers, adds, “We are excited to see the potential of ddMRI being validated through clinical trials, which will pave the way for a new clinical field that will benefit dental practitioners and patients alike.”
Training and Availability
Dentsply Sirona will feature the system at its booth during the American Dental Education Association (ADEA) Annual Session in Montréal, Canada, from March 21–24, 2026.
A two-day, hands-on training program for clinicians will be held in April 2026 at the University of Minnesota. The course will cover imaging protocols, clinical procedures, and image acquisition and interpretation.
Photos: Dentsply Sirona