by Jaimee Morgan, DDS, and Stan Pressley, DDS
How to include bleaching in your protocol
 |
Orthodontic treatment has always provided a challenge when it comes to keeping the teeth and gingiva healthy. Clinicians and patients alike have struggled with plaque buildup, gingival irritation, and the ensuing decalcifications around orthodontic brackets and bands. With approximately 80% of orthodontic patients falling into the preadolescent and adolescent age groups, oral hygiene is especially difficult based on their emotional-development stage. This age group is known for rejecting authority, being influenced by peer pressure, and believing that because of their uniqueness they will be exempt from compromised health.1 This includes decalcification and the long-term adverse effects of poor oral hygiene.
Conventional home care methods of oral hygiene include disclosing tablets, brushing (whether mechanical or manual), oral irrigating devices, mouth rinses, fluoride rinses, topical fluoride, and flossing. Even with all this armamentarium, it still comes down to motivation and compliance. While conventional in-office oral care methods do not rely so much on patient compliance, patients generally have these done only twice per year and therefore, the long-term benefits are not truly significant. More emphasis must be placed on home care with parental involvement.
 |
| Jaimee Morgan, DDSo |
 |
| Stan Presley, DDS |
Recently introduced products that have shown long-term benefits include fluoride-releasing bonding agents, fluoride-releasing band and bracket adhesives, fluoride varnishes, and antimicrobial orthodontic hardware (such as OrthoShield Safe-T-Tie Ligatures from Ortho Organizers, Carlsbad, Calif). Even with these stellar products, the most effective course of action still remains the age-old approach of attacking the problem at its source: hence, a combination of repeated home care instruction, frequent prophylaxis, and in-office and home-delivered topical fluoride. The patient is not released from participation in this effort.
Key Ingredients
Carbamide peroxide, hydrogen peroxide, and tray-delivered fluoride—although excellent in decreasing plaque, inflammation, decalcification, and the incidence of decay—are often overlooked. This omission is probably due to the common misconception that these materials must be used with custom trays that, in an orthodontic setting, could impede the planned movement of the teeth—or as the teeth moved, the trays would no longer fit. New delivery systems with prefilled, semicustom, disposable trays address this dilemma.
Carbamide peroxide was introduced in the late 1960s as an oral antiseptic. It has been well-demonstrated to be both safe and effective in reducing plaque, inflammation, and decay.2-11 Carbamide peroxide breaks down into hydrogen peroxide and urea. The concentration of hydrogen peroxide it breaks down to is approximately one third of what the carbamide peroxide started out as.
For example, 10% carbamide peroxide breaks down to approximately 3% hydrogen peroxide and 7% urea, while 15% carbamide peroxide breaks down to 5% hydrogen peroxide and 10% urea. Carbamide peroxide’s whitening effects were discovered by accident in the late 1960s by an orthodontist (William Klusmier) using removable appliances containing carbamide peroxide to improve the health of the gingival tissue. Once its whitening effects were emphasized in an article in 1989 that described the use of a custom tray delivery system, carbamide peroxide’s other benefits were overshadowed.12
The benefits of fluoride are well-known: It is lethal to bacteria; it aids in the remineralization of enamel; it forms fluorapatite, which is more acid-resistant than hydroxyapatite; and it has long-term desensitizing effects. Fluoride’s desensitizing effects come from its ability to block the tubules and slow the flow of fluid that causes sensitivity.13 Potassium nitrate is also a desensitizing agent, and it has been used in toothpastes (such as Sensodyne from GlaxoSmithKline US) for many years. Potassium nitrate easily penetrates the tooth similarly to hydrogen peroxide and has a direct effect on the nerve. It interrupts the pain cycle by preventing the nerve from repolarizing after it has depolarized.13 It is used primarily for acute sensitivity. The recent addition of fluoride and potassium nitrate to hydrogen peroxide and carbamide peroxide bleaching agents has created a renewed interest in the ability of bleaching agents to improve the health of the teeth. Besides decreasing the incidence of sensitivity from bleaching, research has shown an improvement in the microhardness of the enamel in teeth treated with this combination.14-17
The Combination Approach
Currently, there is only one delivery system that provides carbamide peroxide, hydrogen peroxide, fluoride, and potassium nitrate together in one product (Treswhite Classic from Ultradent Products, South Jordan, Utah). This prefilled, adaptable, disposable tray has a low-concentration carbamide peroxide gingival barrier and a 9% hydrogen peroxide gel in the tray with potassium nitrate and fluoride. The trays adapt to and around the orthodontic appliance. Because of the high concentration of hydrogen peroxide, the patient is instructed to wear each tray only 30 to 60 minutes per day. Nine percent hydrogen peroxide is approximately equal to 30% carbamide peroxide. The patient will benefit from the whitening effects of this concentration, but the other benefits may not be as noticeable because of the limited contact time. Since a longer application of these materials appears to be more beneficial, a combination approach may be desirable. Lowering the percentage of hydrogen peroxide allows more contact time of all the ingredients with the teeth and gingival tissues, thereby amplifying their favorable effects.
Two new products that provide potentially improved health of the teeth and soft tissues are prefilled, adaptable, disposable trays of fluoride (Flor-Opal from Ultradent Products) or potassium nitrate and fluoride (Ultra-Eze from Ultradent Products). These trays will adapt to and around the orthodontic appliances, and each one can be worn up to 90 minutes, at which time the materials become inactive. Although these materials may provide a healthier situation, they do not necessarily provide the motivation factor that was missing before their use. Once again, patient compliance must be relied upon.
Appearances Matter
Research has shown that physical attractiveness and self-esteem are directly related.18-20 For the patient or the patient’s parent, the driving force behind a great deal of orthodontic treatment is aesthetic enhancement rather than functional improvement. This information is the motivational key in convincing your patients to participate in “cleaning up their act,” so to speak.
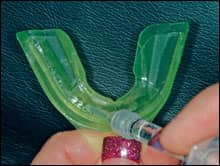 |
| Figure 1: The patient fills the tray. |
Let your patients know that periodically you will be whitening their teeth during their orthodontic treatment. Suggesting bleaching during orthodontic treatment creates interest because most of them will want to know what will happen in the areas where the brackets are. Once it is explained to them that carbamide peroxide and hydrogen readily penetrate the teeth and that those molecules go in all directions, even under the brackets, that initial interest turns to motivation.
Instead of using the fluoride and/or potassium nitrate trays mentioned above alone, have patients add a 10%, 15%, or 20% carbamide peroxide gel containing additional potassium nitrate and fluoride (PF) to the trays prior to inserting them into their mouth. The whitening component provides the motivation for the patient, while the other ingredients have been shown to improve gingival health and increase the microhardness of the enamel as well as decrease potential for sensitivity during the bleaching treatment. Although the fluoride and/or potassium nitrate that lines the prefilled trays becomes inactive approximately 90 minutes after insertion into the mouth, the sustained release carbamide peroxide with PF is active for approximately 8 to 10 hours. Have the patient increase the wearing time and reap the benefits of a prolonged exposure to the ingredients.
Case Presentation 1
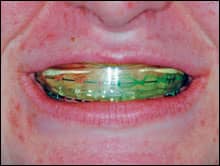
A 22-year-old orthodontic patient who had been experiencing difficulty with her home care regimen was an ideal candidate for a combination treatment using potassium nitrate, fluoride, and carbamide peroxide. She saw the value in the whitening effects, but the clinician was looking forward to the benefits to the gingival tissue and the teeth. The patient was instructed to remove the prefilled, adaptable potassium nitrate and fluoride tray from the package. She was then asked to syringe a thin ribbon of the 10% carbamide peroxide with PF between the ribbons of PF that were already in the tray (Figure 1, above right). After placing the tray into her mouth, she was to gently adapt the colored outer tray to her teeth and then remove it, leaving the thin, clear inner tray (Figures 2 and 3, below). She was informed that the longer the tray could stay in place, the faster the whitening effect would take place. She was given a 10-day supply of both the prefilled trays of PF and the syringes of 10% carbamide peroxide with PF.
 |
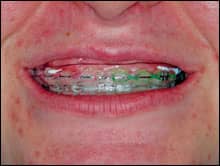 |
| Figures 2 and 3: After the patient places the tray, she removes the colored outer tray, leaving the inner tray. | |
In-office or “power bleaching,” as some call it, can be done by hygienists in most states. In-office bleaching may not provide the longer-lasting benefits of the trays containing PF and carbamide peroxide and/or hydrogen peroxide, but it may be used as a reward for those patients who are exhibiting excellent oral hygiene. The hydrogen peroxide will bleach under the brackets without affecting the existing bond strengths in the same way that the above-mentioned trays do. Once the in-office bleaching procedure is completed, place a prefilled tray of potassium nitrate and fluoride to decrease the potential for sensitivity and add some of the other benefits that PF provides. A side benefit of both in-office and home bleaching methods is that they remove stains from around and on porcelain brackets.
Case Presentation 2
A 12-year-old female patient had been in orthodontic treatment for several months when her mother inquired about the possibility of whitening her teeth during treatment. Her oral hygiene habits were fair, and the decision to bleach was based on information reported by Scherer et al.21 In his bleaching study, patients appeared to be motivated in their oral hygiene home care. With the patient fully reclined, the orthodontic wire was removed, the patient’s lips were moistened with lip balm, and then the teeth were isolated using a self-retaining cheek retractor. A combination bite block/tongue retractor was then placed, and the patient was asked to bite on it, keep the tongue lingual to it, and maintain that position throughout the bleaching treatment. The gingival tissue was then air-dried, and a resin gingival barrier was syringed onto the gingival margins of the teeth to be bleached, overlapping approximately 1 mm onto the teeth. The barrier was also extended one tooth beyond where the bleach was to be placed. Special attention was paid to the interproximal tissues to prevent lingual migration of the bleach. After placement, the resin barrier was light-cured. The gingival barrier replaced the need for a rubber dam, saving time and making the procedure more comfortable for the patient.
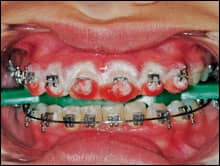
After isolation was completed, the chemically activated 38% hydrogen peroxide bleaching agent was mixed using a syringe-to-syringe method. To show how the bleaching agent travels once it penetrates the tooth as well as how effective it is, it was syringed only onto the incisal half of the teeth (Figure 4). The gel was kept off the gingival barrier to prevent accidental contact with the soft tissue. The gel was left on the teeth for 15 minutes, removed, and reapplied, for a total of two cycles. Normally, a four-cycle regimen of 15 minutes each would have been used, but due to the patient’s youth, it was decided that two cycles would be enough for one treatment. Surgical suction was used to remove the gel, with no rinsing of the teeth between applications. After removal of the second application, the teeth were thoroughly rinsed and high-volume suction was used. The gingival barrier was teased away from the tissues with an explorer, followed by another thorough rinse. It has been reported that the adverse effects of bleaching on bond strengths lasts approximately 7 days, so it is crucial to wait at least that long after completion of bleaching before performing any bonding treatments, including repositioning brackets.22
 |
 |
| Figure 4: The bleaching agent is only on the incisal. Figure 5: The maxillary teeth are clearly whiter. | |
Three weeks after the bleaching treatment, the patient returned to the office for evaluation of the bleaching effects. A bracket was removed to show that there was no dark area where the bleach did not have direct contact with the surface of the tooth. When the maxillary bleached teeth were compared with the mandibular unbleached teeth, a significant whitening effect was obvious (Figure 5).
It is important to remember not only the health issues associated with patients undergoing orthodontic treatment, but also their aesthetic needs during and after treatment. Appealing to their desire for aesthetic enhancement may be one of the most effective means of improving the health of the soft tissues and protecting the teeth by increasing the microhardness of the enamel, making them less impervious to decay. Using at-home tray delivery of prefilled adaptable disposable trays or providing in-office treatments are excellent and easy methods of getting your patients to “clean up their act.”
Jaimee Morgan, DDS, practices cosmetic and restorative dentistry in Salt Lake City. She was heavily involved in research in association with the Division of Esthetic Dentistry at the University of Texas for several years. She has presented numerous scientific papers, has had several articles published in the leading dental journals, and lectures internationally on aesthetic dentistry from a combined research and clinical approach. She is an instructor at the University of Utah in the General Practice Residency Program. She can be reached at
Stan Presley, DDS, practices cosmetic and restorative dentistry with his wife, Jaimee Morgan, in Salt Lake City. He was one of the founding members of the South Texas Chapter of the AACD, where he served as secretary and vice president. He is codeveloper of the Simplified Layering Technique for composites. He lectures internationally and has contributed numerous articles demonstrating realistic and learnable procedures for general practitioners.
References
- Proffit WR, Fields HW Jr, Sarver DM. Contemporary Orthodontics, 4th ed. St Louis, Mo: Mosby Elsevier; 2007:66.
- Zinner DD, Duany LF, Chilton NW. Controlled study of the clinical effectiveness of a new oxygen gel on plaque, oral debris and gingival inflammation. Pharmacol Ther Dent. 1970;1:7-15.
- Zinner DD, Duany LF, Llorente M. Effects on urea peroxide in anhydrous glycerol on gingivitis and dental plaque. J Prev Dent. 1978; 5(1):38-40.
- Fogel MS, Magill JM. Use of an antiseptic agent in orthodontic hygiene. Dent Survey. 1971;October:50-54.
- Shipman B, Cohen E, Kaslick RS. The effect of urea peroxide gel on plaque deposits and gingival status. J Perdontol. 1971;42:283-285.
- Tartakow DJ, Smith RS, Spinelli JA. Urea peroxide solution in the treatment of gingivitis in orthodontics. Am J Orthod. 1978;73:560-567.
- Shapiro WB, Kaslick RS, Chasens AI, et al. The influence of urea peroxide gel on plaque, calculus and chronic gingival inflammation. J Periodontol. 1973;44:636-639.
- Reddy J, Salkin LM. The effect of a urea peroxide rinse on dental plaque and gingivitis. J Periodontol. 1976;47:607-610.
- Sherer W, Boylan R, Bhatt S. Vital bleaching agents and oral antiseptic: Effect on anaerobic bacteria. J Esthet Dent. 1992;4(3):84-85.
- Haywood VB, Heymann HO. Nightguard vital bleaching: how safe is it? Quintessence Intl. 1991;22(7):515-523.
- Haywood VB. Nightguard vital bleaching: Current concepts and research. J Am Dent Assoc. 1997;128(Supplement):19S-25S.
- Haywood VB, Heymann HO. Nightguard vital bleaching. Quintessence Intl. 1989;20:173-176.
- Haywood VB. A comparison of at-home and in-office bleaching. Dentistry Today. 2000;19(4):44-53.
- Al-Qunaian TA. The effect of whitening agents on caries susceptibility of human enamel. Oper Dent. 2005;30(2):265-270.
- Clark LM, Barghi N, Summitt JB, Amaechi BT. Abstract 0497. Presented at: Annual Meeting of the American Association of Dental Research; March 10, 2006; Orlando, Fla.
- Basting RT, Rodrigues AL, Serra MC. The effects of seven carbamide peroxide bleaching agents on enamel microhardness over time. J Am Dent Assoc. 2004;135(5)556-558.
- Amaechi BT, Clark LM, Barghi N, Summitt JB. Enamel fluoride uptake from fluoridated carbamide peroxide bleaching gel. Abstract 0498. Presented at: Annual Meeting of the American Association of Dental Research; March 10, 2006; Orlando, Fla.
- Patzer GL. Reality of physical attractiveness. J Esthet Dent. 1994;6(1):35-38.
- Patzer GL. Self-esteem and physical attractiveness. J Esthet Dent. 1995;7(6):274-277.
- Patzer GL. Understanding the causal relationship between physical attractiveness and self-esteem. J Esthet Dent. 1996;8(3):144-147.
- Scherer W, Palat M, Hittelman E, Putter H, Cooper H. At-home bleaching system: Effect on gingival tissue. J Esthet Dent. 1992;4(3):86-89.
- Morgan-Godwin JA, Barghi N, Berry TG, Knight GT, Hummert TW. Time duration for dissipation of bleaching effects before enamel bonding. J Dent Res. 1992;71:abstract 590.










