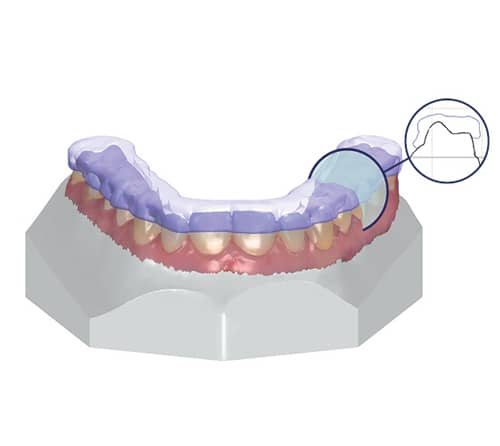
3Shape Ortho System enables orthodontic professionals to overlay DICOM, cephalometric, and 2D pictures along with intraoral scans for orthodontic case analysis and planning, treatment simulations, and the design of FDA-cleared orthodontic appliances.
The FDA now extends Ortho System’s 510(k) market clearance to include the design of dental retainers, splints, mouthguards, and nightguards, in addition to previously cleared indications: custom metal bands, export models, and indirect direct bonding transfer media.
3Shape will present its Ortho System at the AAO Annual Session in San Diego on April 22, 2017, at 10 am. The event will take place at the company’s AAO booth.
This FDA market clearance also includes 3Shape Indirect Bonding and transfer media solutions, as well as several appliance design workflows that guide the user through the design phase and relevant production parameter settings.