by Jackie Dorst
Sterilizer options for orthodontic instruments

Instrument reprocessing and sterilization must reflect the same high level of technology and efficiency as the office computer systems do. Too often, I visit orthodontic practices and see antiquated sterilization rooms outfitted with old equipment that has been used for 20 years or more.
What message does an antiquated sterilization system send to your patients? Do you proudly give a tour of your sterilization area during the new-patient tour? Have you had patients question instrument sterility?
Patients expect to see the same type of sterile instrument package opened for orthodontic treatment that they see when they visit their dentist. If patients and parents do not see a sterile package, then questions or doubts will arise in their minds about instrument safety and infection-control procedures. Patient concerns about instruments could even influence treatment acceptance for parents with a high awareness of disease transmission and the standard precautions taken by other health care providers.
Patient perception and changing regulations and guidelines from the Centers for Disease Control and Prevention (CDC) and state Dental Boards are prompting orthodontists to remodel and update equipment for orthodontic instrument reprocessing. Instrument sterilizers used in orthodontic offices must have the approval of the Food and Drug Administration (FDA) for use as a medical instrument sterilizer. For example, it is a violation of the State Dental Practice Act to use a convection oven for instrument sterilization, because kitchen devices are not approved by the FDA for use in health care facilities.
The Complete Package

Packaging the instruments after sterilization is not acceptable. When consulting with orthodontists, I have observed instruments being placed in peel-vue pouches after sterilization. The pouches have an external chemical indicator that turns a color after sterilization. Imagine what patients who are knowledgeable about instrument sterilization would think when they observe a nonsterile instrument package used for patient treatment. They would know that the package was not sterile because they would see that the indicator color was unchanged.
Unsaturated Chemical-Vapor Sterilizers
Unsaturated chemical-vapor sterilizers use a proprietary formula of alcohol and 23% formaldehyde to generate the chemical vapor that sterilizes the instruments. Because there is lower humidity in a chemical-vapor sterilizer than there is in a steam sterilizer, usually it is less corrosive to carbon steel instruments. Instruments must be dry before they can effectively be sterilized using this method.
Potential exposure to formaldehyde is a health concern for orthodontic team members processing instruments. Some state Occupational Safety and Health Administration (OSHA) programs require chemical monitoring of formaldehyde exposure in the sterilization area. Also, requirements for disposal of spent solution should be reviewed with state and local authorities.
Dry-Heat Sterilizers
Dry-heat sterilizers have traditionally been used to sterilize orthodontic pliers. There are two types of dry-heat sterilizers: static air and forced air. Static-air sterilizers have heating coils in the sides and bottom of the chamber. The hot air rises through natural convection. This method of heating is slow, requiring 1–2 hours for sterilization. Forced-air dry-heat sterilizers circulate heated air throughout the chamber at a high velocity. Forced-air dry-heat sterilization reduces the time required to sterilize instruments to 12 minutes at 375°F.
If the sterilizer is loaded up with lots of instruments, however, the thermostat senses that the temperature is not adequate for sterilization and thus keeps increasing the temperature. I have observed readings of 415°F during sterilization cycles. These extremely high temperatures scorch and burn packaging materials, and cause mirrors to break. These temperature are harmful to handpieces and ultrasonic inserts, and can even damage pliers over time.
All sterilizers have a weight limit that is used to indicate how many instruments can be effectively sterilized in each load. Overloading a sterilizer is one of the most frequent causes for failing a spore check (a test done with a biological monitor). Overloading can also cause damage to the sterilizer and the instruments.
Because of the high temperatures in dry-heat sterilizers, it is very difficult to package instruments in pouches or wrap cassettes for dry-heat sterilization. The high temperatures can scorch or burn the paper and plastic packaging materials designed for use with autoclaves and chemical-vapor sterilizers. The only instrument-packaging material designed for use in dry-heat sterilizers is high-temperature nylon, which is available as self-sealing pouches or tubing that can be heat-sealed. Before you buy either of these, make sure that the package states that the clear nylon material is designed for use in dry-heat sterilizers.
As discussed previously, dry heat has traditionally been used to sterilize orthodontic pliers because it is considered less corrosive than “wet” forms of sterilization and does not dull the cutting edges of pliers. In recent years, however, orthodontic instrument manufacturers are producing higher-quality orthodontic pliers of surgical-grade stainless steel that are guaranteed to withstand all methods of sterilization, including autoclaves. Orthodontic practices purchasing new instruments should ask the manufacturer about recommended methods of cleaning and sterilization, and what is covered under the guarantee for the instruments.
Autoclave Sterilizers

There are two categories of autoclaves approved by the FDA for use in the United States: gravity and prevacuum autoclaves.

At the end of the sterilization cycle, gravity autoclaves automatically “crack” the door open for drying. The cracked-door drying is slow, and frequently results in wet or slightly moist instrument packages. Wet packages have the potential to wick microorganisms through the packaging material. Additionally, sharp instruments are more likely to puncture a wet package than a dry one, and, of course, residual moisture is not at all good for orthodontic instruments.
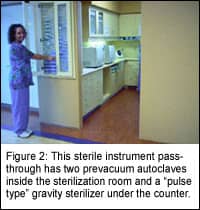
Prevacuum autoclaves, on the other hand, introduce steam into the sterilizer’s chamber faster and more effectively than gravity autoclaves by using a vacuum pump to remove the air before introducing the steam into the chamber. These sterilizers also have the advantage of a postvacuum drying cycle that yields crinkly, dry packages. The sterilization and drying cycles are both completed with the door closed, so there are no more wet packages to deal with when the door of the sterilizer opens.
The use of a vacuum pump reduces the prevacuum and postvacuum autoclave cycle time 40% over gravity autoclaves. Instrument cycling time—the time it takes to clean, dry, package, and sterilize instruments—is a challenge for busy orthodontic offices. While the new prevacuum autoclaves are faster than gravity autoclaves, the prevacuum autoclave chambers are only 10 inches in diameter. There is a larger prevacuum sterilizer on the market, but it requires 1 hour for a sterilization cycle and the cabinets must be specially designed to accommodate the large size. The 10-in chamber holds only four large orthodontic cassettes.
Orthodontists who use large instrument cassettes must decide whether to purchase two gravity autoclaves with 11-in chambers or two to three prevacuum autoclaves with 10-in chambers. The chamber size is not as significant if the office is using pouches rather than cassettes.
When I’m consulting with an orthodontist on sterilization design, the first decision we have to make is which size cassettes will be used. Only after choosing the proper cassette size can the correct size and number of sterilizers be determined.
Instrument sterilization is going high tech. Are you ready?
A state-of-the-art sterilization center features instrument cassettes, recessed ultrasonic cleaners, hospital-type sterilizers with closed-door sterilization cycles, and lighted sterile instrument storage. Letting your patients see sterile, packaged instruments stored in a sterile pass-through cabinet is a visual reassurance of just how clean and safe your orthodontic office is. z
Jackie Dorst presents practical continuing-education courses on infection control, sterilization design, and OSHA. She is the owner of Safe Practice, located in San Clemente, Calif, a sterilization consultancy, and is on the board of directors of the Organization for Safety and Asepsis Procedures. She can be contacted at [email protected] or (949) 366-9170.
Recommended Reading
1. Centers for Disease Control and Prevention. Guidelines for infection control in dental health-care settings—2003. Morbidity and Mortality Weekly Report Recommendations and Reports. 2003;52(RR-17):1-61.
2. Occupational Safety and Health Administration. Final Rule on Occupational Exposure to Bloodborne Pathogens. 1991;56:64004.
3. Organization for Safety and Asepsis Procedures. Infection Control in Practice: New Instrument Reprocessing Technologies. 2005;4(1):1-5.
4. Organization for Safety and Asepsis Procedures. From Policy to Practice: OSAP Guide to the Guidelines. Available at: www.osap.org.